Gastrointestinal Stromal Tumors Treated by Mesylate Imatinib to the Teaching Hospital of Brazzaville
Context: The significant advanced into the comprehension of some mechanism of carcinogenesis of GIST and, thanks to the program GIPAP (Glivec International Patient Assistance Program); our patients should benefit from the minimum standard fare gratuitously of the mesylate imatinib treatment since 2008.
Objectives: To evaluate the treatment of patients followed by GIST imatinib mesylate into the Service of Carcinology and Radiotherapy of Brazzaville Teaching Hospital.
Materials and Methods: It was done a descriptive study with retrospective incorporating 11 cases over a 66 months period from 1st January 2010 to 30th June 2015. Patients were treated with a combination of surgery and targeted therapy (imatinib mesylate 100 mg 2cp x 2/day) or exclusively by targeted therapy. Overall survival was calculated by the Actuarial method.
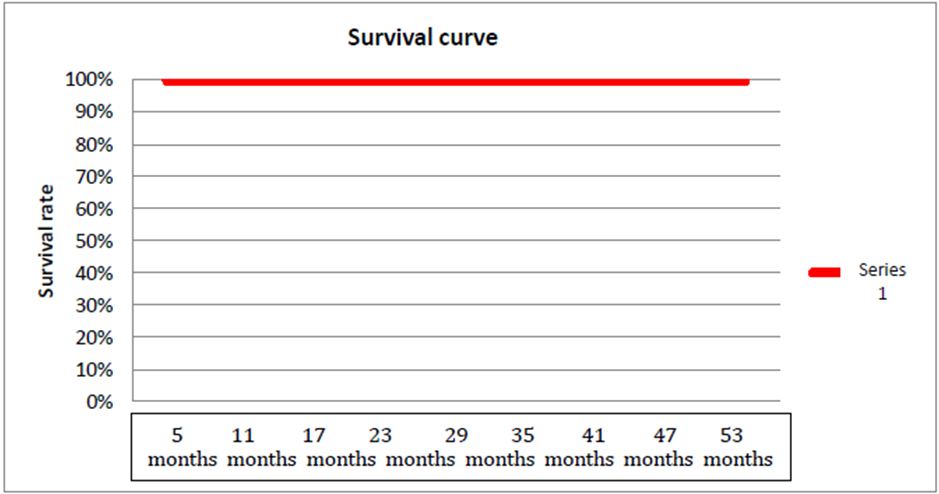
Results: The middle age within the two sexes was 54 years with the extremes from 42 to 69 years. A male predominance was noted. On average consultation time was 8 months with extremes of 2-8 months. The upper gastrointestinal bleeding was the main symptom. The GIST was with gastric location more of 80% of cases. Nine patients benefited from the surgery and in adjuvant the imatinib; 2 patients have in exclusive way benefited from the imatinib. As for my Actuarial method, the survival to 53 months is of 100%.
Conclusion: Therapy progress indisputable brought these last years with the use of the imatinib into the GIST metastasis, also his effectiveness, showed after the surgery into the high risk of relapse, just like his excellent profile of tolerance. All the medical decisions around of that kind of patient are to be considerate in the Meeting of Multidisciplinary.
Keywords: Results; Therapeutic; GIST; Imatinib Mesylate; GIPAP
The Gastrointestinal Stromal Tumors (GIST) of Anglo-Saxons is the more frequent mesenchymatous tumors of digestive tract [1]. Deriving from Cajal cells, and placed in the interface between the myocytes and the ending of nervous plexus of digestive tract. In Congo, the first case was brought in 2002 [2]. A targeted therapy inhibited the KIT; the imatinib has modified the prognosis, of advanced form of GIST [3]. We bring our experiment, after the imatinib administration in the GIST.
The Service of Carcinology and Radiotherapy of Brazzaville Teaching Hospital was very useful for us for working. The GIPAP project offers the imatinib gratuitously to the following patients and countries below: the Republic of Congo, the Democratic Republic of Congo (DRC), the Centre Africa Republic and the Republic of Chad.
The patients were the carriers of that compliant; they signed a lighted consent form by GIPAP.
The data have been collected from the individual medical records and the form of the anatomopathologic report as well as the immunohistochemistry presented by the patients.
We used the telephone as an aid in order to contact the sick persons. We also used an inquiry pre-established form to receive the data.
• Type and period of study: It was done a descriptive study with retrospective over a 66 months period from 1st January 2010 to 30th June 2015.
• Inclusion criteria: Have been included, all patient of gastrointestinal stromal tumor carrier with the immunohistochemistry proof (positive with the tracer CD117 and/or CD34); and having benefited from imatinib treatment.
•Non-inclusion criteria: Within this study, we did not include all patients who are a carrier of an intestinal stromal tumor without having the immunohistochemistry proof (positive with the tracer CD117 and/or CD34); having not benefited from imatinib treatment or having been treated in exclusively by the surgery.
• Variable studies: These are what we studied:
- Age : at the moment of diagnostic and starting the treatment
- Sex: sex never changes but we must precise the gender.
- General state : before and after the treatment estimate by the Performans Status (PS) of WHO
(according to ECOG «Easten Cooperation Oncology Group» side from 0 to 4);
- Clinical signs, anatomic location;
- Size of tumor, tumoral extension;
- Microscopic aspect : (fusiform and epithelium) ;
- Immunohistochemistry : (positivity CD117 and/or CD34) ;
- Therapeutic and results, side effects, the survival.
The patients have been treated just by the mesylate imatinib or associated to the surgery.
These were some surgical technics ways:
✓ An atypical gastrectomy: also known by wedge gastrectomy in the tumors of the big curve.
✓ An inferior polar gastrectomy: it was a partial gastrectomy of 3/4 or of 4/5 from the stomach with security
margin in healthy tissue of small gastric curve.
✓ A hemicolectomy and a splenectomy.
We did administer the imatinib at the dose of 400 mg/day within the oral use in one hold in all cases. The evaluation of the answer under Imatinib according to the criteria referred as «Choi» and for the tumor answer according to the RECIST (Response Evaluation Criteria In Solid Tumor) [4].
• Statistical analysis
The data have been captured using EpiData 3.1 software
The statistical analysis has been done from EpiData 3.1 software and EPI INFO 3.5.3.0.
The global survival rate has been calculated according the Actuarial method.
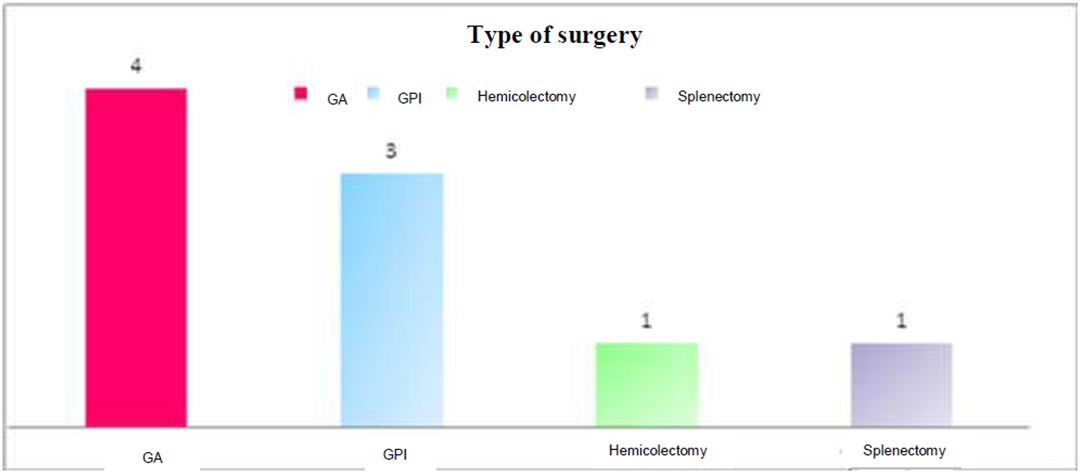
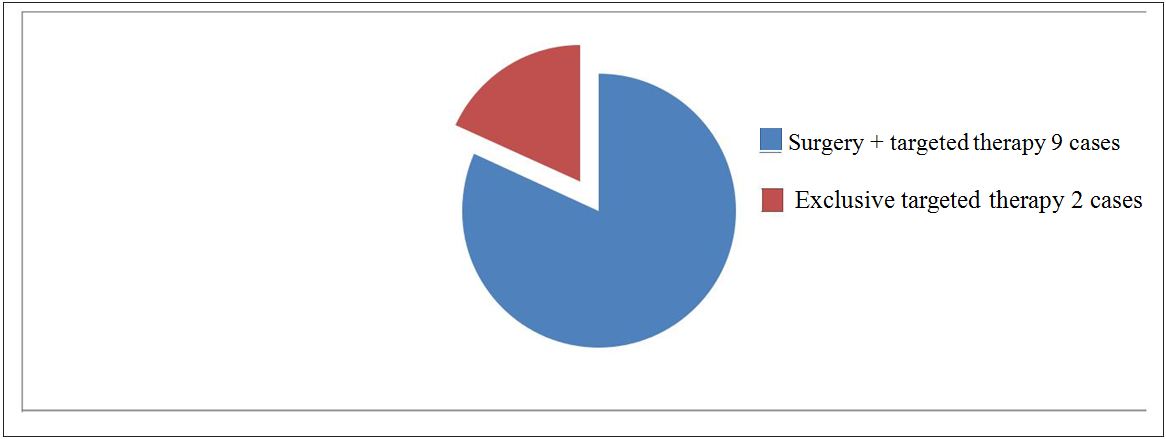
During the study period, we recorded 13 cases of GIST, which 9 cases treated by the surgery and targeted therapy in adjuvant, 2 cases in exclusive way by the targeted therapy. Two other cases treated by the surgery only them which were excluded from the study.
The GIST represents 2.16% on the whole gastrointestinal malignant tumor which have been diagnosed during the study period. That corresponds to 0.02% of office hours in the Service of Carcinology and Radiotherapy of Brazzaville Teaching Hospital.
The characteristic of GIST are presented in the synoptic board ( Table 1).
The median age within the two sexes was of 54 years with the extremes from 42 to 69 years.
We noted 8 men and 3 women with a sex ratio of 2.66.
The socio-demographic characteristics are presented in the board ( Table 2)
We could notice a blood pressure patient and of diabetes.
The middle deadline of office hours was of 8.18 (±8.13) months with the extremes from 2-28 months.
The middle deadline of diagnosis was of 2.54 (±1.57) months with the extremes from 1-5 months.
Within the 6 cases, the patients have consulted for high digestive hemorrhage, within 3 cases for abdominal pain and within 2 cases for abdominal mass.
Table 2 shows the development of PS sign before and during the mesylate imatinib treatment.
On the 11 patients, the high endoscopy has been fulfilled in 8 cases, the pelvic abdominal sonogram and the abdominal tomo-densitometry in 4 cases and thoracic in 2 cases; the thorax X-ray has been carried out in 9 cases. The different locations observed from afar were hepatics (02 times), then hepatics and peritoneum (02 times).
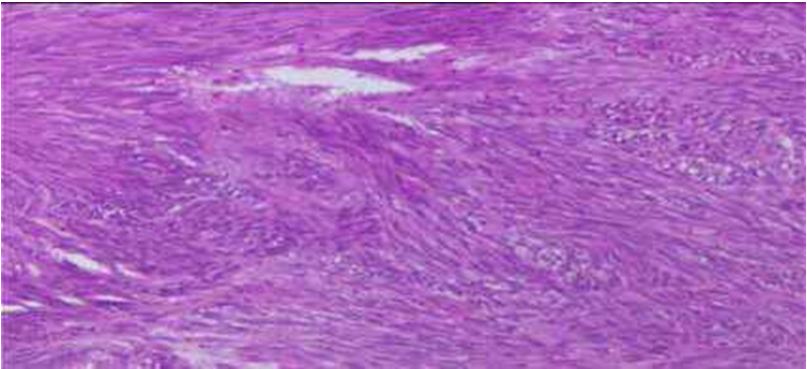
The stomach was of the primitive location in 9 cases on 11 and the median size of 8 cm with the extremes going from 2 to 19 cm. It was of GIST fusiform in 9 cases and the index about mitosis was high in 7 cases.
The anatomopathologic characteristics are presented in the board ( Table 3)
A biopsy was carried out in 8/11 cases and it was negative in 5 cases on 8. The diagnosis has been installed on the operating room in 9/11 cases and in 2 cases by the laparoscopy diagnosis.
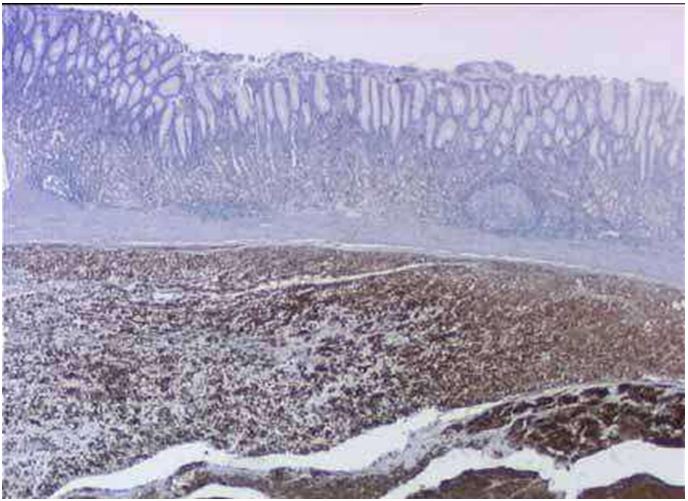
All the patients were greatly positive to CD117 and CD34.
One patient has benefited from the molecular biology on 11 cases and, disintegration to the level of the exon 9 of KIT gene; also, we could identify an absence mutation of PDGFRA gene.
The Figure 1 and 2 represent the microscopic aspects and the immunohistochemistry KIT (CD117) and CD34 tracing.
The surgical treatment (atypical gastrectomy, partial gastrectomy, inferior polar gastrectomy, hemicolectomy, splenectomy) has been followed of the treatment by the imatinib in 9 cases and in 2 cases an exclusive administration of imatinib.
Seven patients on eleven have benefited from the macroscopic R0, on which, one 1 case of extra-digestive GIST.
The Figure 3 presents the different types of treatment. The Figure 4 represents different types of therapeutic means fulfilled.
Targeted therapy: The side effects were an edema of inferior limbs in 2 cases, a palpebral edema in 2 cases, a cramp and an abdominal pain respectively in 1 case.
Table 4 represents the distribution according to the type of answer
Table 5 represents the different characteristics of GIST.
Survival data is represented in the Table 6
Survival curve of patients according to the Actuarial method is represented in the Figure 5.
Our study was about to bring the therapeutic results of GIST under of the imatinib in the frame of GIP AP project at Brazzaville Teaching Hospital.
The limits of that study were essentially the size of the sample and the research of exon KIT and/or exon PDGFRA by molecular biology, which his interest is both prognosis and therapeutic in the GIST.
A means of judgment is situated in the evaluation criteria of responses to the treatment, especially in the GIST non-re-divisible or immediately metastatic. That means could be corrected by the global survival of our series.
The GIST is the rare tumors of digestive tract [5]. We could not ourselves think the incidence in this study; for, it is the study of cases stem from the hospital data. Yet, we can consider that it is about a rare pathology, representing 2.16% of cancers of digestive tract and 0.02% of consultations to the Carcinology and Radiotherapy Service of Brazzaville Teaching Hospital.
According to Mouaquit, et al., the GIST represents 0.1 to 3% of all digestive cancers and 1 to 3% of gastric tumors [6]. That rarity is found in the African series: they represent 2% of the whole of gastrointestinal malignant tumor and 0.06% of hospitalization; then, 13 cases in twelve years; 40 cases in fourteen years [7,8,9]. On the contrary, in the North of Europe and in Asia, the incidence rate varying between 11 and 14.5 by millions of inhabitants have been brought while in the North of America the incidence rate are situated between 6.8 and 9.1 by millions of inhabitants [7]. However, their frequency is likely underestimate in our structures owing to the absence of equipped laboratory to the technics of immunohistochemistry and the molecular biology [10,11].
The median age in our series was of 54 years with the extremes of 42 to 69 years, that is superior to which brought in Nigeria 46 years, but definitely inferior to those who decry in Europe, in United State and in China that they are among 63 – 69 years [8-11]. The male predominance observed in our study is hard to be interpreted on the result of the size of our sample. The published series in Africa present the same limits; for, they are mono-centric and with small size [8,12,13]. In the great occidental series and Asian, there is no male predominance clearly proved, the results which were brought are often discordant, but the sex-ratio is generally similar of 1 [10,14].
The most revealing symptoms are a digestive bleeding which is let out and pains or an abdominal gene non-specific; rarely occlusive syndromes of digestive perforations, the discovery of metastasis by a majority hepatitis is the most seldom abdominal pain (3 cases), and the abdominal mass which is let out (2 cases). Several authors have admitted the fact [7,15-17].
The PS sign which is altered (PS ≥ 1 in the 11 cases) before the treatment, has been improved (PS 0 in 7 cases, PS 1 in 4 cases) observed during the treatment. Our results are similar to those brought by Tatsuo, et al. [18]. The high endoscopy permitted to display the lesions and to precise the headquarters, the biopsy were negative to the most of our patients as described in the literature [4,19].
In accordance with the brought data, the stomach remains the elective headquarters of GIST [10-12]. The tumors are diagnosed to an advanced stage to our patients; 4 patients were metastasis during the diagnosis, only one sick person presented a 2 cm tumor.
Fall et al., bring 6 cases of metastasis on 10 patients, and 7 patients on 10 presented a palpable abdominal mass during the diagnosis [7]. Moreover, in the European series, American, the diagnosis is made to an early stage [10,11,14].
As described in the literature, there is a predominance of cells of fusiform types in our series which has been met [20,21].
The GIST is characterized in our series by the constant positivity to the tracer CD117.
That immunohistochemistry characteristic, is similar to the data already published by diverse authors, which show a good sensitivity of tracer CD117 for the GIST diagnosis with positivity’s rates between 80 and 95% [8,11,22]. The CD34 tracer, equally positive to all our patients, is nevertheless less sensitive [22,23].
Many authors think that the curative treatment of GIST is based on the complete surgery without the entering of tumor [3,16]. Nine patients have benefited from the surgery on which we could see from afar two patients of metastasis, seven other have benefited from the macroscopic R0. The complete resection rates are better in the occidental and Asian series [14,22]. Recurrence cases appear often in spite of the complete macroscopic [7,8]. For Weber,et al. [24]. These one could be locals or from afar of the primitive tumor but they are most of all intra-abdominal and about the liver and the peritoneum in the most of cases, suspecting a possible resistance.
In our study any case of recurrence has been recorded under imatinib after a median deadline of 29 months. This could be justified by the adjuvant treatment systematic by the imatinib to all the patients operated, the risk of recurrence being evaluated to each patient in post-operative according to the character of Fletcher et al. [25]. Moreover, Miettinen et al. stress the importance of location, opposing the gastric GIST of better prognosis and the non-gastric GIST (small intestine, rectum) [17].
The inoperable tumor prognosis or recurring is considerably improved since the use of the imatinib, an inhibitor of tyrosine-kinase which blocks the trans-membrane tyrosine-kinase mutated at the level of fixation site of ATP. Thus, administered to the dose of 400 mg in a daily hold; this allows to obtain a local control of GIST in 80% of cases [9,26]. In certain cases, it could improve the results of the surgery when it is administered in neo adjuvant or adjuvant [3,13]. In our student, all benefits from treatment by the mesylate imatinib, despite of its prohibitive cost thanks to the GIPAP program. The effectiveness of imatinib is currently established, the daily dose recommended in the framework of AMM is of 400 mg by day, given until progress, intolerance or refusal of sick person [17].
The median survival of our patients is comparable with those of the surgical series [3].
However, the weak rate of complete answer at the imatinib and the existence of secondary resistances make that outside of non-re-divisible GIST, metastatic or recurring, the initial surgery must remain the treatment rule of GIST [7,16].
The GIST is mesenchymatous tumors the most frequents of digestive tract. Their diagnosis is based on the morphologic aspect and the immunohistochemistry co-expression of bio-tracers KIT and/or CD34. Consequently, the efforts ought to be made to equip our laboratories anatomy of pathology in KIT of immunohistochemistry and the molecular biology.